Rhubarb Poison Information
The Rhubarb Compendium
[Home]
[Recipes]
[Previous page]
[Next page]
13 - Poison Information
Rhubarb contains oxalate, which have been reported to cause poisoning when large quantities of raw or cooked leaves are ingested.
13.1 - The poison in rhubarb
Oxalates are contained in all parts of rhubarb plants, especially in the green leaves. There is some evidence that anthraquinone glycosides are also present and may be partly responsible. It is not clear as to the exact source of poisoning from rhubarb, possibly a result of both compounds. The stalks contain low levels of oxalates, so this does not cause problems.

13.2 - Chemical Composition of Rhubarb
| --- | --- | Water | 91.2 - 96.1 % | Solids, total | 4.0 - 8.48 % | Solids, soluble | 2.1 - 3.0 % | Sugars | 0.3 - 2.3 % | Nitrogen, total | 0.127 - 0.211 % | Proteins | 0.6 | Fats | traces - 0.7 % | Pectins (Ca-pectate) | 0.11 - 0.77 % | Tannins | 0.058 - 0.110 % | Fiber, Crude | 1.1 - 1.2 | Carotenes | 1.2 ug/100g | Ash | 0.62 - 1.23 % | Acids | --- | --- | Malic | 0.73 - 2.15 % | Oxalic acid | 0.124 - 1.360 % | Citric acid | 0.07 - 0.18 g/100g | Acetic acid | 0.02 - 0.16 g/100g | Fumaric acid | 0.01 - 0.10 g/100g | Glucolic acid | 0.01 g/100ml | Succinic acid | 0.02 g/100ml | pH | 3.01 - 3.59 | Ash constituents mg/100g |
During World War I rhubarb leaves were recommended as a substitute for other veggies that the war made unavailable. Apparently there were cases of acute poisoning and even some deaths. Some animals, including goats and swine, have also been poisoned by ingesting the leaves. 21,37
The biodynamic (toxicity) mechanism by which oxalic acid works is somewhat different from organic poisons and is more analogous to heavy metal poisoning. Organic poisons often work through at the biochemical level, e.g. cyanide by interfering with respiration at the cellular level, strychnine by screwing up intersynaptic transmission. There are many molecular substances in foods which offer no nutritional benefit, and must be processed and excreted. Oxalic acid, for example, is excreted in the urine, and its crystals are commonly found in microscopic urinalysis. Too much oxalic acid in the urine will result in kidney or bladder stones. Calcium combines with oxalic acid to form the less soluble salt, calcium oxalate, which is also found in kidney stones. Plant leaves, especially rhubarb, cabbage, spinach, and beet tops, contain oxalic acid. Oxalic acid is also found in potatoes and peas. Vitamin C is metabolized to oxalic acid; it contributes to over-saturation of the urine with crystals and possibly to stone formation. 54,55
13.3 - More about Oxalic acid
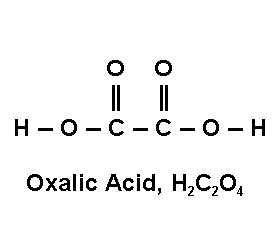
Oxalic acid is a strong acid of the composition HOOC-COOH, which crystallizes as the ortho-acid (HO)3 CC (OH)3 . It is sometimes also called “ethane diacid”. It occurs naturally in some vegetables (like rhubarb). The can also be produced by heating sodium formate and treating the resulting oxides with sulfuric acid. It can also be obtained by the action of nitric acid on sugar, or of strong alkalis on sawdust. The product is normally traded as colorless crystals with a melting point of 101.5?C, and can be dissolved in water or alcohol. Oxalic acid reduces iron compounds, and is therefore used in metal polishes, stain removers, and writing inks. When it absorbs oxygen, it is converted to the volatile carbon dioxide and to water, and it is used as a bleaching agent, in detergents, and as a mordant in dyeing processes.59

A Rhubarb Leaf
13.3.1 - How toxic is rhubarb?
| Oxalic Acid % | ||
|---|---|---|
| Leaf blade | Petiole | |
| Water soluble | 0.46 - 0.51 | 0.23 - 0.32 |
| Water insoluble | 0.13 - 0.21 | 0.16 - 0.22 |
| Total | 0.59 - 0.72 | 0.39 - 0.54 |
From an MSDS (Material Safety Data Sheet) for Oxalic acid, LD50 (LD50 is the Median Lethal Dose, which is the dose of a drug or chemical predicted to produce a lethal effect in 50 percent of the subjects to whom the dose is given) in rats is 375 mg/kg. So for a person about 145 pounds (65.7 kg) that’s about 25 grams of pure oxalic acid required to cause death. Rhubarb leaves are probably around 0.5% oxalic acid, so that you would need to eat quite a large serving of leaves, like 5 kg (11 lbs), to get that 24 grams of oxalic acid. Note that it will only require a fraction of that to cause sickness. 57,58,60,61
13.3.2 - Symptoms of Oxalic Acid Poisoning
On the body body as a whole one might experience weakness, burning in the mouth, death from cardiovascular collapse; on the respiratory system - difficulty breathing; on the eyes, ears, nose, and throat - burning in the throat; one the gastrointestinal system - abdominal pain, nausea, vomiting, diarrhea; and on the nervous system - Convulsions, coma. 56,58

Rhubarb plant
13.3.3 - Precautions for rhubarb gardening
- Trim leaves from stalk immediately.
- Don’t use stalks from frost bitten plants.
- Wash the stalks well.
- Children should be taught to eat only the rhubarb stalks, preferably under supervision
13.3.4 - New stuff added
CALCIUM CARBONATE (precipitated chalk) For removing excess acid from juices. Used extensively to remove oxalic acid from Rhubarb juice.63
Readers of The Rhubarb Compendium may be interested in…
William R. James, Wilma Roberts James, J. Carroll Reiners (Illustrator), Arla Lippsmeyer (Illustrator)
This book describes and illustrates sixty-five species of poisonous plants. Fifteen additional pages of another seventy-five species are included with brief descriptions and illustrations. It’s not necessary to remove all these plants from our gardens, we can still enjoy them for their beauty, fragrance, medicinal uses and aesthetic value. Know Your Poisonous Plants is a highly recommended addition to any private or public gardening book collection. [b15]
Footnotes
-
a USENET posting From: Barry_Carlson@mindlink.bc.ca (Barry Carlson), Subject: Rhubarb, Date: Thu, 20 May 1993 01:39:06 GMT, Part of Lysator Academic Computer Society at Lysator.
-
- Rheum rhaponticum poisoning, http://res.agr.ca/cgi-bin/brd/poisonpl/, from: Agriculture and Argi-Food Canada Research Server
-
- USENET POSTING: Arthur A. Simon, Jr. aasimon@ios.com posted to Newsgroup rec.food.cooking on Mon Jul 29 19:46:03 1996
-
- GLOSSARY OF TERMS AND SYMBOLS USED IN PHARMACOLOGY, http://med-amsa.bu.edu/Pharmacology/Programmed/glossary.html
- MSDS for OXALIC ACID, gopher://atlas.chem.utah.edu/11/MSDS, University of Utah, Chemistry dept
-
59.Oxalic Acid (HOOC.COOH).2H2O, http://www.metalogic.be/MatWeb/reading/acids/e_acoxal.htm, METALogic N.V. on-line materials information system
-
- USENET POSTING: andywlms@esslink.com (Andy Williams) posted to Newsgroup rec.food.cooking on 24 Jul 1996 18:47:47 GMT
-
- USENET POSTING: lhom@nature.berkeley.edu (Louis Hom) posted to Newsgroup rec.food.cooking on 25 Jul 1996 00:26:12 GMT
-
- Guide to Plant Relationships (for food allergy and intolerance identification), http://www.purr.demon.co.uk/Food/RelatedPlantList.html, This is a list of plant groups, primarily food plants, intended to assist people with food intolerances and allergies.
-
- WINEMAKING INGREDIENTS, http://www.tek-efx.com/wine_ingred.html
[Home]
[Recipes]
[Previous page]
[Next page]
[About]
[Contribute]
© RhubarbInfo, 1996-2003
Updated on September 1, 2004
Email: dan at rhubarb info dot com
 Know Your Poisonous Plants
Know Your Poisonous Plants